Introduction to Ussing
Product Summary
- For the study of transport across epithelial membranes
- Most widely used equipment in the field
- Can be used for electrophysiological measurements (Isc, Rm), diffusion-based measurements, or both
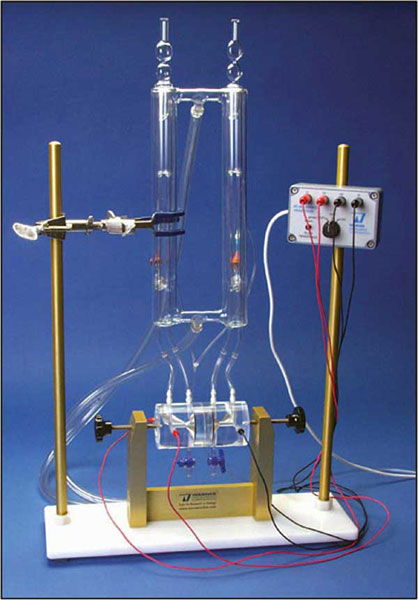
An Ussing System, used to measure transport across epithelial membranes, is generally comprised of a chamber and perfusion system, and, if needed, an amplifier and data acquisition system. The heart of the system lies in the chamber with the other components performing supporting roles. The ‘Classic’ chamber design, first introduced by the Danish physiologist Hans Ussing in the early 1950’s, is still in wide use today. However, several newer designs are now available that optimize for convenience and for diffusion- or electrophysiology-based measurements.
Epithelia are polar structures possessing an apical (or mucosal) and basolateral (or serosal) side. It is the movement of electrolytes, non-electrolytes, and H2O
Ussing systems have been used to make measurements from native tissue including stomach, large and small intestine, gall and urinary bladder, skin, and trachea, as well as from tissue derived cell monolayers from various sources including renal tubes, pancreas, and salivary and sweat glands. A well designed Ussing chamber supports an epithelia membrane or cell monolayer in such a way that each side of the membrane is isolated and faces a separate chamber-half. This configuration allows the researcher to make unique chemical and electrical adjustments to either side of the membrane while maintaining complete control.
Study types
Ussing systems can be used for either electrophysiology- or diffusion-based studies, or for a combination of both. They can also be used for radiotracer studies wherein the ionic species under transport is radiolabled. For convenience, we will consider radiotracer studies to operate within the context of a diffusion- or electrophysiology based system.
While each approach described above makes use of a chamber and fluid handling system, an electrophysiology-based setup has the added dimension of a current and/or voltage clamp amplifier and data acquisition system. Relevant components include:
| Diffusion-based system | Electrophysiology-based system |
|---|---|
|
|
A systems approach
Warner Instruments offers a wide variety of Ussing systems embodying different design philosophies. Fortunately, the various components are generally interchangeable allowing for a great degree of flexibility and customization.
The goal of this article is to outline the advantages and disadvantages associated with each Ussing chamber design with an eye towards guiding you in selecting the best components for your application.
Ussing measurements
A basic understanding of different Ussing measurements commonly made can guide you in determining which components best fit your needs when building a system.
As stated earlier, Ussing systems can be defined as being either diffusion- or electrophysiology-based. A diffusion-based system is generally focused on measuring transepithelial fluid transport and is used to follow net movement of H2O or solute across the membrane. By itself, diffusion-based systems do not provide specific information regarding the underlying transport mechanism and are best suited towards measurements of leaky epithelia characterized by electroneutral transport.
While fluid transport can result in a volume change in the recording chamber, it is usually quantified by measuring a corresponding change in a volume marker. Volume markers include changes in salt or dye concentration, or changes in physical characteristics such as fluid capacitance or resistance. Advantages of fluid transport measurements using a volume marker include good temporal resolution and sensitivity to small fluxes (volume changes as low as ±1 nl/min have been reported). A disadvantage is the requirement for small volume chambers.
An electrophysiology-based system focuses on measuring transepithelial electrical responses to experimental perturbations. These systems are used to quantify the operation of electrogenic pathways in the membrane (e.g., ion pumps, channels, etc). As such, an electrophysiology-based system carries the additional hardware requirement of a voltage and/or current clamp amplifier, a data acquisition system, and collection/analysis software.
Basic measurement parameters in electrophysiology-based Ussing systems include transmembrane voltage (Vt), epithelial membrane resistance (Rt), and short circuit current (ISC). ISC
A limitation with these systems is that non-electrogenic ion transport mechanisms such as fluid transport and electroneutral ion transport cannot be directly monitored. This limitation, however, can be addressed by employing indirect or secondary measurements such as ion replacement, transport inhibition, and the use of hormones and second messengers.
The use of radioisotope tracers is one measurement technique deserving special mention. This technique can be applied equally well to both diffusion- and electrophysiology-based measurements and is usually employed to provide information regarding ion-specific transport mechanisms. For example, a diffusion-based model cannot identify the fluid being transported or if the measured volume change is the result of a hydrostatic or osmotic process. Finally, if an osmotic-driven volume change is mediated by an ionic mechanism, then the responsible ion is not identified. By comparison, a limitation with the electrophysiology-based model is that while ionic transport can be measured, the specific ion crossing the membrane is not determined. This is especially true for multi-ionic salt conditions. For both cases, the use of a radiolabeled ionic species allows for directly monitoring ion-specific translocation for the two systems described here.
The components
An Ussing system is comprised of a chamber and perfusion system, and if needed, an amplifier and data acquisition package. If working at defined temperatures, then a circulating water bath is also needed to thermoregulate the chamber system water jackets.
The chamber system
While decisions need to be made for each step in assembling a complete Ussing system, the greatest complexity is associated with choosing the chamber and its associated tissue support. Once these elements have been addressed, then the task of selecting the other components is straightforward.
All Ussing chambers have the following features in common: a means to hold the membrane while minimizing tissue damage, a means for holding and exchanging solutions, a means to facilitate precise electrode placement, and a means to control solution temperature and gas load.
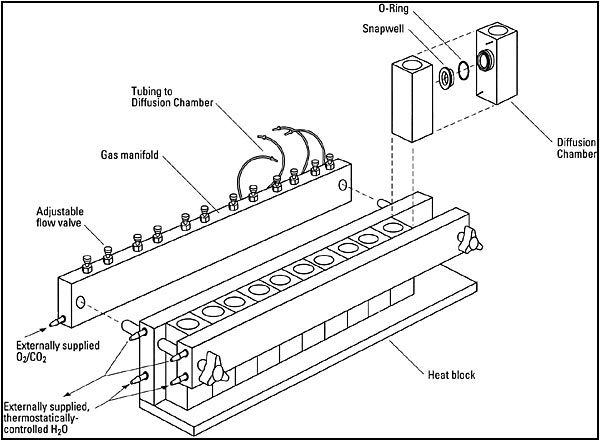
Ussing chamber systems from Warner include the Self-Contained (incorporating all the elements of the classic setup within a small housing), and the Navicyte horizontal and vertical systems (allowing simultaneous measurement from many epithelia in parallel). The Self-Contained chambers make use of inserts allowing them to accommodate different tissues as well as cultured cells on permeable supports (e.g., Snapwell, Millicell, or Transwell).
The Self-contained chamber system functions the same as the Classic system as designed by Hans Ussing in the 50's, albeit with a smaller volume footprint and with supporting the tissues via inserts, which is a different approach than that taken by Dr. Ussing. This system also supports cultured cells on Snapwell, Millicell, or Transwell culture cups.The Multiple channel chamber systems are comprised of the Navicyte Vertical and Navicyte Horizontal designs. Both make use of a support assembly to hold anywhere from 1 to 6 independent chambers and to facictate thermal regulation vua a water jacket, and to couple the perfusion and electronic components to the chambers.
- The Navicyte Horizontal configuration accommodates from 1 to 6 chambers and is ideal for studying mucosal layers at an air/liquid interface. Navicyte Horizontal chambers make use of a mounting ring to support the tissue of interest or a Snapwell culture cup for cells.
- The Navicyte Vertical configuration is particularly well designed for diffusion-based studies and accommodates from 1 to 6 chambers. Tissues are supported directly in the chamber block and no insert is required. However, for cultured cells, a chamber block supporting a Snapwell culture cup is also available.
Electrophysiological measurements
If making measurements of membrane resistance (Rm) or short-circuit current (ISC), then an amplifier and data acquisition system is also required. We offer complete data acquisition systems for 2, 4, 6, or 8 channels under the UssingChart moniker that include the amplifiers, digitizer, and software. Detailed information can be seen on the UssingChart webpage.
References
- Liddle’s syndrome mutations increase Na+ transport through dual effects on epithelial Na+ channel surface expression and proteolytic cleavage
Knight KK, Olson DR, Zhou R, Snyder PM
Proc Natl Acad Sci U S A. 2006 Feb 21;103(8):2805-8 - A Simple, Cross-linked Collagen Tissue Substitute for Corneal Implantation
Liu Y, Gan L, Carlsson DJ, Fagerholm P, Lagali N, Watsky MA, Munger R, Hodge WG, Priest D, Griffith M
Invest Ophthalmol Vis Sci. 2006 May;47(5):1869-75 - Culture of murine nasal epithelia: model for cystic fibrosis
Grubb BR, Rogers TD, Diggs PC, Boucher RC, Ostrowski LE
Am J Physiol Lung Cell Mol Physiol. 2006 Feb;290(2):L270-7
Related Products
Related Files
Customers from Europe and Australia can use the Multi Channel Systems - Online Shop to order Warner products or create quotes.
| Order No. | Model No. | Product Description | USA Price | Order |
|---|